Translate this page into:
Periodontal health is the key for overall health
*Corresponding author: Ruchi Srivastava, Department of Periodontology, Saraswati Dental College, Lucknow, Uttar Pradesh, India. drruchi117@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Srivastava R, Chandra C, Jha V, Dubey A, Sharma T, Goel T. Periodontal health is the key for overall health. Asian J Oral Health Allied Sci 2022;12:3.
Abstract
Periodontal diseases affect about 20–50% of the population worldwide. Periodontitis is the infection of the tissues that areis initiated by plaque deposition along with anaerobic microbiota, for example, Actinobacillus actinomycetemcomitans, Bacteroides forsythus,_and_Porphyromonas gingivalis. The repercussion of plaque deposition is calculus formation which augments inflammation. This inflammation coaxes to mislay of periodontal ligaments and alveolar bone which terminates mobility and tooth loss. This critique summarizes the effects of these periodontal maladies on oral health and overall health. Many studies have concluded and proven the relationship between periodontal maladies and systemic diseases such as cardiovascular diseases, nephropathy, diabetes, osteoporosis, and respiratory disorders. Substantial attempts are made in the critique to show the pathological mechanism between oral maladies and systemic diseases. This review represents that periodontal treatment could ameliorate systemic diseases and highlights the attention of health-care specialists to emphasize the need for an association between dental and medical disciplines.
Keywords
Periodontal diseases
Periodontal medicine
Systemic disease
INTRODUCTION
Functional dentistry and oral health ramifications longevity are notable for a long time. In Japan, on New Year’s Feast, people eat HAGATAME MOCHI (teeth strengthening rice cake) to wish for longevity and strengthening teeth and bones. The oral cavity is the arrefour of medicine and dentistry and also a window to the overall health. The oral cavity encounters unabating challenges of varied oral complications of systemic malady and disorders. The main aim of the critique is to emphasize how oral hygiene is the key to periodontitis and periodontitis is the key to oral health and overall health and the uniqueness of periodontal infection is that it is among the most prodigious human infections. The cardinal justification for this uniqueness is the unwonted anatomic hallmark that the tooth progresses through the integument so that portions of it are bared to the external environment while a portion is strongly rooted in the alveolar bone. Incongruity to the exteriors of most parts of the human body, the superficial layers of the dente does not shed and thus furnish a relatively unswerving surface for microbial colonization.[1] This expedites microbes maintaining contiguous immediate propinquity to the periodontal tissues. Forbye, the tooth surface, and the affixed microorganisms are imbued in an aqueous environment, where any baktron infection is barely able to be controlled by the puissant mechanisms of the host immune responses and the antimicrobial therapy.
PERIODONTAL DISEASES
Periodontal diseases are multifactorial inflammatory diseases of the periodontium (supporting structure of teeth) originated by a specific or group of specific microbiota that terminates in the destruction of periodontal ligament (PDL) and alveolar bone with the formation of periodontal pocket and gingival recession or both.[2]
SIGNS AND SYMPTOMS OF PERIODONTAL DISEASES
Halitosis (bad breath)
Red and swollen gums
Tender and bleeding gums
Painful chewing
Tooth mobility
Sensitive tooth
Receding gums and longer appearing teeth
Loss of stippling
Furcation involvement
Clinical attachment loss (CAL)
Pocket formation
Food impaction.
PATHOGENS AND PATHOGENESIS
The ubiquitous Gram-negative bacterial genera in the rima oris include Treponema, Prevotella, Bacteroides, Porphyromonas, Capnocytophaga, Actinobacillus, Peptostreptococcus, Fusobacterium, and Eikenella. Earlier investigations identified Actinobacillus actinomycetemcomitans, Porphyromonas gingivalis, and Tannerella forsythia as causative doers in periodontitis and much of the exploration on periodontitis perpetuates to focus on these microorganisms. However, neoteric investigations have determined that the rima oris contains approximately 500–700 prevalent species of microbes; this microbial community is designated as the oral microbiome.[3] The oral microflora is present on the gingival epithelium, in saliva, and other surfaces of the rima oris and is also intensive in dental plaque. The dental plaque is a marshaled biofilm of microorganisms; either affixed to the tooth surface or to the other microbes to resist and survive antibiotic treatment or the host defense mechanisms. As the biofilm thrives, microbial dysbiosis transpires and causes a progressive shift from Gram-positive to predominately Gram-negative anaerobic species, ensuing in biofilm formation underneath the gingival surface. In addition, sucrose metabolism by the oral plaque biofilm leads to the formation of organic acids, which portrays a crucial role in pH diminution and demineralization of the dental surface.[4] Thus, frequent sucrose consumption is also notorious to impel dysbiosis of the supragingival microflora, aiding the development of dental caries.
PERIODONTITIS AND ORAL HEALTH
Oral health remits to the health of our oral cavity. It is much more than clean teeth. It involves the gingiva and supporting tissues, lining of the mouth and throat, the palate, tongue, salivary glands, muscles of mastication, lips, nerves, and bones of the upper and lower jaw. The oral malady is a major health encumbrance for many realms and affects individuals throughout the lifetime. It is a silent epidemic. Periodontitis is one of the biggest threats to oral health, the other being dental caries. Periodontitis affects the tissues that both support and surround the tooth. According to the CPITN developed by Ainama et al., 1982, healthy periodontium connotes the absence of calculus, no gingival bleeding on probing and periodontal pockets.[5] If left untreated, periodontitis can lead to tooth loss, in adults. There is pathological detachment of periodontal fibers from the cementum, the junctional epithelium migrates apically, and bone loss is evident. It also leads to the pocket formation and CAL which can expose the tooth root to the oral environment leaving them prone to dental caries (root caries). Bone resorption can lead to osseous defects, tooth mobility, and eventually, tooth loss making the individual edentulous depriving him of adequate nutrition. Bone resorption also makes bone fragile and more prone to fracture. Pockets lead to food impaction and debris collection which again forms the source of bacterial colonization and these bacteria can enter the pulp chamber through lateral foramina and apical foramen (perio-endo lesion) leading to pulpitis and pulpal necrosis which can further lead to a chain of problems if left untreated; for example, pulpitis can lead to an abscess, granuloma, cyst formation, sinus formation, osteomyelitis, cellulitis, and space infections.
In 1879, Willoughby D. Miller proposed that oral bacteria can antecedent various diseases in the organs other than the oral cavity in an article “THE HUMAN MOUTH AS A FOCUS OF INFECTION.”[6] In 1898, Hunter probed and contemplate the term “oral sepsis” (Hunter, 1900) and consider it for causing various systemic diseases. Further, in 1912, Frank Billings[7] posits “THEORY OF FOCAL INFECTION” that tells about the nature and spread of the infection.
RELATIONSHIP BETWEEN PERIODONTAL AND SYSTEMIC DISEASES
Periodontitis a threat to stroke
Cardiovascular diseases (CVD) embrace a group of maladies such as CHF, CAD, VHD, and stroke. Amidst these, atherosclerosis forms the paramount component of CVD’s and is the major cause of demises worldwide.
Atherosclerosis fosters by the deposition of the multiple atherosclerotic plaques within the innermost walls of the arteries (hardening and furring of arteries).[8,9] The denouement leads to coronary thrombosis, MI, and stroke. Increasing evince explains the interrelation and supports the role of inflammation and infections, which plays a pivotal role. Several mechanisms that could elucidate this association have been probed [Figures 2 and 3]. Host riposte to periodontal pathogens may kindle the production of inflammatory mediators such as TNF-α, PGE-2, IL-1ß, IL-6, and C-reactive protein which boost the burgeoning of the pre-existing atheromatic plaques and effectuates increased number of cardiovascular ailments. Furthermore, periodontal pathogens have the caliber to actuate atheroma formation and platelet aggregation.

- Relationship between periodontal and systemic diseases.
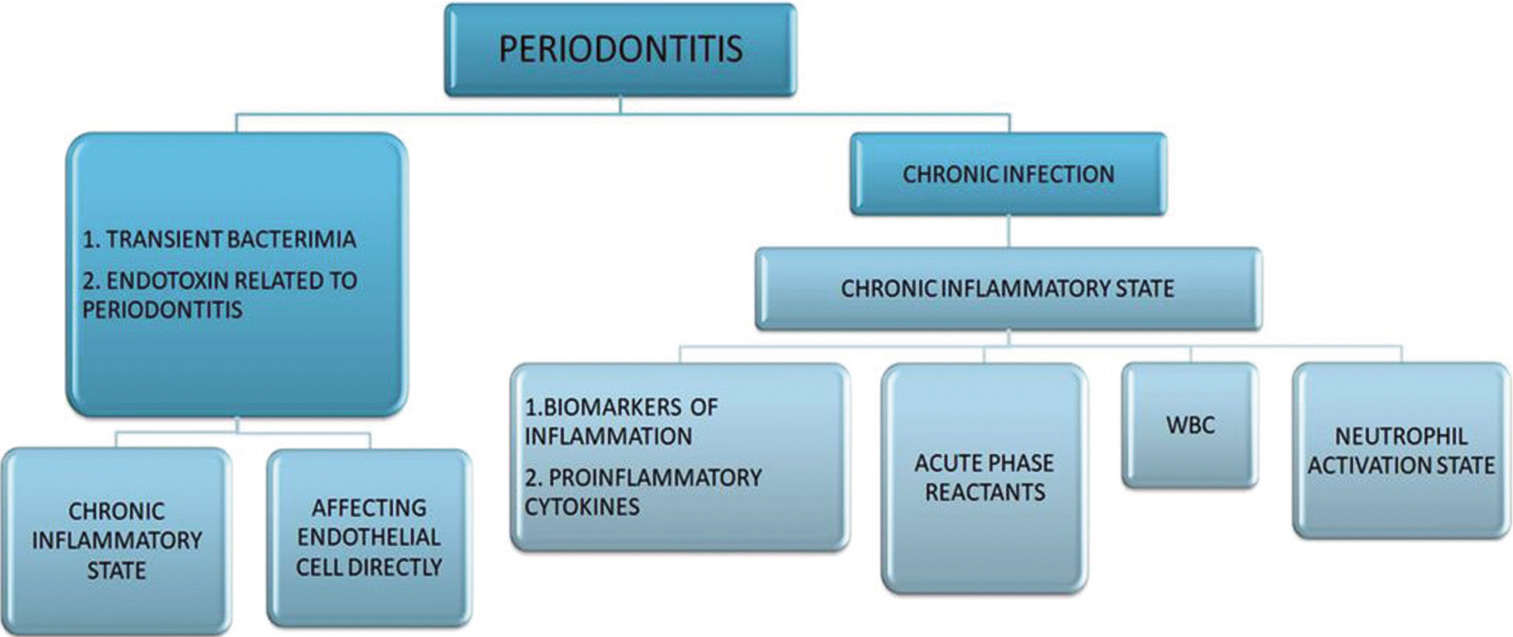
- Mechanism elucidation association between periodontitis and cardiovascular diseases.
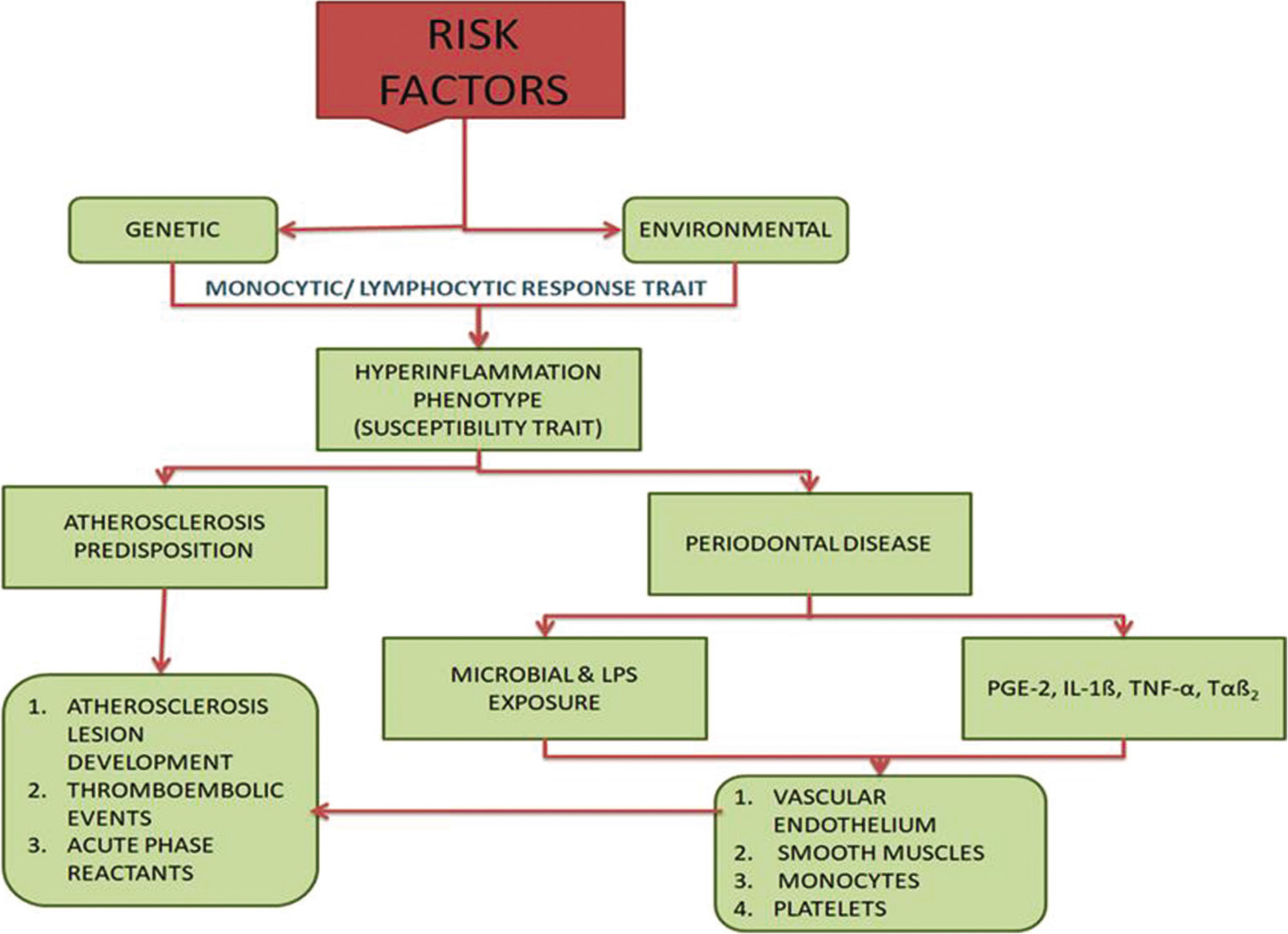
- Biological tenability; risk factors of cardiovascular diseases.
MI consorts with acute viral and bacterial infections and is interrelated to periodontitis.[8,10] Studies of atheroma during endarterectomy divulged that more than half of lesions embodied periodontal pathogens which may lead to thrombosis, rupture of atheroma, and hypercoagulability. Indigent dental health is a menace for cerebrovascular ischemia. Elevated markers of inflammation consorted with periodontitis are also themselves the menace for the stroke. The entrenched risk factors do not portray fully the risk of stroke [Figure 3]. Dental ministrations and intensive treatment mitigated the risk of stroke.
PERIODONTAL DISEASE AND DIABETES MELLITUS
Diabetes is a type of disease delineated by hyperglycemia which upshots either due to insulin secretion deficiency and/or turndown in insulin action. It is unambiguously confirmed as a risk factor for periodontitis. In a diabetic individual, there is an approximately 3-fold increase in the risk of periodontitis compared with non-diabetic individuals, and on the other hand, periodontitis has also boosted the mechanism of diabetic mellitus. Hence, this emphasized a bidirectional relationship between diabetes and periodontitis. Several investigations had been carried out to underpin this hypothesis in the Gila River Indian Community, which proposed that severe periodontitis can be cognate with an increased peril of poor glycemic control.[11] In another longitudinal study, Demmer et al. investigated non-diabetic individuals for 5 years and found increased HbA1c levels. Periodontal disease is contemplated as the sixth complication of diabetes.[12] The biological pathway that connects diabetes and periodontitis includes inflammation, immune responses, cytokine biology, and neutrophil mechanism. Several meta-analyses have appraised that effective periodontal treatment can improve glycemic control. In 2008, a meta-analysis involving 485 individuals had established a decrease of glycated hemoglobin (HbA1c) by 0.46% following periodontal treatment.[5] In 2010, another meta-analysis study including 371 individuals had also established a decrease of HbA1c by 0.40% afterward the periodontal therapy.[13]
PARADIGM FROM PERIODONTITIS TO RESPIRATORY DISEASE
Malady affecting the respiratory system embracing lungs, bronchi, pleural cavity, trachea, and upper respiratory tract is entitled as RESPIRATORY DISEASES. Rima oris is incessant with the trachea and is a portal for the colonization of respiratory pathogens. The upper respiratory tract is contaminated with microorganisms procured from the oral, pharyngeal, and nasal regions. Menace of certain pulmonary diseases increases by elevated bacterial encumbrance due to atrocious oral hygiene and periodontitis. Respiratory pathogens colonize the dental plaque which when aspirated into the upper airway reaches the lower airway and clings to the alveolar epithelium.[14] It transpires most substantially in high-risk patients for instance intensive care unit patients, nursing home residents, older subjects, and hospitalized patients (requiring ventilator). Periodontitis alters the pathogen’s adherence to the pulmonary epithelium. Alveolar bone loss accessorizes with the risk of COPD. Smoking acts as an “EFFECT MODIFIER” in a correlation between periodontitis and COPD. Tweaked oral hygiene reduces the rate of pneumonia in high-risk patients.
PERIODONTITIS AND PREGNANCY
During pregnancy, women who develop periodontitis (estimated one in five) have a greater probability of adverse pregnancy outcomes. About 11.1% estimated preterm delivery rate is associated with periodontitis. Other problems other than these are preeclampsia, low birth weight, and being small for gestational age.[15] The hypothesized mechanism of action shows that periodontitis leads to inflammatory response and endothelial dysfunction. Periodontal pockets are the reservoir of oral microbiota. Studies show increased Campylobacter rectusin pregnant women and red complex bacteria such as P. gingivalis and Prevotella intermedia.
PERIODONTITIS AND NEPHROPATHY
In end-stage renal disease patients on hemodialysis (HD), periodontitis may be a benefactor to systemic inflammation. HD patients unveil an inordinate number of periodontal pathogens and also evince interconnection between hypoalbuminemia and severe periodontal disease. The proffered mechanism for the corollary of periodontitis on the development of renal disease is systemic inflammation. Periodontal pathogens adhere, assail, and proliferate in coronary endothelial cells concocting atheroma and impairing the vasculature relaxation. Periodontitis employs corresponding effects on the vasculature of nephrons. CKD and CVD allocate umpteen risk factors. Both are cognate with CRP and chronic inflammation analogous with periodontitis which further emanates to endothelial dysfunction.[16-18]
SKIN DISEASES
The occupancy of butterfly rashes has gesticulated systemic lupus erythematosus, but this also suggests dermatological disorders. In the patients with butterfly rashes, the pervasiveness of oral malady is 7.9%.[6] These rashes would emanate bilaterally on the malar region, bridge of the nose and chin. An article titled as “periodontal disease in association with megakaryocytic thrombocytopenia” and systemic lupus erythematosus (SLE) had delineated first case in the dental literature proclaiming a 17-year-old female with severe periodontal malady cognate with SLE and thrombocytopenia. In 2012, another case report had published a case of 40-year-old female of butterfly rashes with periodontal maladies. Later, both the conditions amend with periodontal therapy.[6]
PERIODONTITIS AND CANCER
Connection between cancer and periodontitis has been established as 24% increase in menace of ensuing cancer has been probed in patients with severe periodontitis. Inflammation is the major facet that relates periodontitis to cancer. Cancer, especially from squamous cells in the area, emulates periodontitis.[19] Long-term inflammation proselytizes tumor growth, development, and progression malignancies which can transpire from areas that are stamped by infection and inflammation as a bit of physiological immune retaliation of the host. The mantle of IL-1ß has also been noted in periodontitis which is also described in tumor invasiveness, tumorigenesis, angiogenesis, tumor host interactions, and metastasis. Correlation between cancer and inflammatory response comprises two mechanisms, namely, exogenous and endogenous.[16] The association is also possible biologically.
PERIODONTITIS AND OTHER INFECTIONS
-
Periodontal diseases result in systemic dissemination of inflammatory molecules which burgeon risk factor for the number of systemic maladies including Alzheimer’s disease (AD). Treponema denticola and certain spirochetes were perceived in patients with AD and the same microbiota is analogous to periodontal diseases. To explicate, the relationship between periodontal disease and AD certain contrivance has been propound.
One stipulates that pro-inflammatory cytokines after systemic dissemination enter blood–brain barrier and cause neuronal damage
Whereas the second proclaims that periodontopathic microorganism can conquer brain directly.
Many cross-sectional and longitudinal studies had divulged that dementia is also a denouement of poor oral health
Enormous epidemiological studies have also revealed the consortium between periodontitis and rheumatoid arthritis
Periodontal maladies and osteoporosis are also omnipresent conditions. A systemic review had proposed the interrelationship between periodontal maladies and osteoporosis with the positive relative radiographical findings.[20]
PERIODONTAL MEDICINE
Conceptualization of periodontal diseases as localized units attacking only the tooth and the supporting structures is oversimplified and needs emendation as they also impact the extraoral health and hence is the key to both oral health and overall health
Periodontal medicine was first appraised by STEVEN OFFENBACHER in 1996,[9] focusing on the plethora of new data substantiating a strong relationship between systemic health and periodontal health. Hence, it is a new discernment into the outmoded problem envisioned.
Periodontitis shows potentiality of dissemination of microorganism intravascularly throughout the body and inclines to be a “SILENT” malady until destruction causes acute symptoms. Periodontitis may behave as a sovereign risk factor or it may exacerbate the existing systemic disease. Hence, dental professionals should broaden one’s horizons and acknowledge oral cavity as one of many intertwined organ systems.
Patient education is precedence and must emphasize on the nature, increased risk and role the periodontal infection would play in systemic diseases. At present, the rationale to medicate the periodontal disease is to safeguard the structure, functions, and the esthetics of dentition which is also important in averting untoward effects on the patient’s overall health.
CONCLUSION
A burgeoning number of evidence in the studies evinces the direct and indirect effect of periodontal microorganisms on overall health. Recent experimental, clinical, and epidemiological studies support the interrelation between inflammation and bacteremia due to periodontal disease, oral and systemic diseases.
We cannot establish an undeviating causal relationship but we can assert very safely with supporting attestation that there is an obvious connection betwixt these biofilms and bacteria detected in the bloodstream. There are compelling epidemiological corroborations but mechanisms are not entirely clarified. More researches are needed to unravel the mechanisms whereby periodontal microorganisms or the ensuing inflammation foster or promote systemic disease. Patient education in this interest is also important. Nonetheless, it is already discernible that the management of periodontitis and proper oral care can definitely impact the mortality, morbidity, and health-care toll associated with oral and systemic diseases. Hence, oral hygiene is the key to periodontitis and periodontitis is the key to oral health and overall health.
Acknowledgement
The authors would like to acknowledge Dr. Vivek Kumar Bains, Professor and Head of Department of Periodontology, Saraswati Dental College, Lucknow (Uttar Pradesh) for his guidance, brilliant comments and suggestions through all the stages of writing this article.
Declaration of patient consent
Patient’s consent not required as there are no patients in this study.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Association between periodontal diseases and systemic diseases. Braz Oral Res. 2008;22:32-43.
- [CrossRef] [PubMed] [Google Scholar]
- Influence of periodontal infection on systemic health. Periodontol 2000. 1999;21:197.
- [CrossRef] [PubMed] [Google Scholar]
- Identification of periodontal pathogens in atheromatous plaques. J Periodontol. 2000;71:1554-60.
- [CrossRef] [PubMed] [Google Scholar]
- Thesis, antithesis, and synthesis in periodontal and systemic interlink. J Indian Soc Periodontol. 2012;16:168-73.
- [CrossRef] [PubMed] [Google Scholar]
- Development of the World Health Organization (WHO) community periodontal index of treatment needs (CPITN) Int Dent J. 1982;32:281-91.
- [Google Scholar]
- Association between systemic lupus erythematosus and periodontitis: A systematic review and meta-analysis. Front Immunol. 2017;8:1295.
- [CrossRef] [PubMed] [Google Scholar]
- Germs, Dr. Billings and the theory of focal infection. Clin Infect Dis. 1998;27:627-33.
- [CrossRef] [PubMed] [Google Scholar]
- Associations between periodontal disease and risk for atherosclerosis, cardiovascular disease, and stroke. A systematic review. Ann Periodontol. 2003;8:38-53.
- [CrossRef] [PubMed] [Google Scholar]
- The association between periodontal diseases and cardiovascular diseases: A state-of-the-science review. Ann Periodontol. 2001;6:9-15.
- [CrossRef] [PubMed] [Google Scholar]
- The relationship between myocardial infarction and periodontitis. Caspian J Intern Med. 2013;4:667-71.
- [Google Scholar]
- A review of the evidence for pathogenic mechanisms that may link periodontitis and diabetes. J Clin Periodontol. 2013;40(Suppl 14):S113-34.
- [CrossRef] [PubMed] [Google Scholar]
- Periodontal status and A1C change: Longitudinal results from the study of health in Pomerania (SHIP) Diabetes Care. 2010;33:1037-43.
- [CrossRef] [PubMed] [Google Scholar]
- Patients' experiences of the impact of periodontal disease. J Clin Periodontol. 2010;37:334-9.
- [CrossRef] [PubMed] [Google Scholar]
- Associations between oral conditions and respiratory disease in a national sample survey population. Ann Periodontol. 1998;3:251-6.
- [CrossRef] [PubMed] [Google Scholar]
- Preterm low birth rate and maternal periodontal status: A meta-analysis. Am J Obstet Gynecol. 2007;196:135.e1-7.
- [CrossRef] [PubMed] [Google Scholar]
- Periodontitis and diabetes: A two-way relationship. Diabetologia. 2012;55:21-31.
- [CrossRef] [PubMed] [Google Scholar]
- Periodontitis and diabetes mellitus: Be true to your teeth. Int Heart J. 2018;59:680-2.
- [CrossRef] [PubMed] [Google Scholar]
- Dental infections as a risk factor for acute myocardial infarction. Eur Heart J. 1993;14:51.
- [Google Scholar]
- Insights into inflammasome and its research advances in cancer. Tumori. 2019;105:456-64.
- [CrossRef] [PubMed] [Google Scholar]
- Periodontal medicine: Impact of periodontal infection on systemic health In: Newman MG, Takei H, Klokkevold P, Fermin A, eds. Carranza: Clinical Periodontology Vol Ch. 18. (10th ed). Philadelphia, PA: WB Saunders and Company; 2009. p. :312-29.
- [Google Scholar]






